Every year, hundreds of thousands of men with low-grade prostate cancer face a recurring dilemma: submit to another biopsy, with its attendant discomfort and small but real infection risk, or hope that the cancer growing quietly inside them hasn’t quietly changed. The dilemma exists because the tools available to guide that decision have, until now, been frustratingly imprecise. MRI scans help. PSA blood tests help. But neither helps enough to let doctors say, with real confidence, that a biopsy can safely wait.
A new study suggests a simple urine test could break that impasse. Evaluated across 11 urology practices in the United States, the test outperformed MRI on every meaningful metric, and in doing so raised a question that researchers are only beginning to take seriously: how many of the biopsies performed on men with low-risk prostate cancer are actually necessary?
The answer, at least in this study, is roughly a third. Maybe fewer.
The Logic of Watching and Waiting
The condition at the centre of all this is called active surveillance, and it represents something of a philosophical shift in how oncologists think about cancer. For decades, a prostate cancer diagnosis meant treatment, often aggressively so, and the side effects of surgery and radiation (incontinence, erectile dysfunction, the whole grim catalogue) were considered the price of being thorough. Then came the evidence, accumulating slowly through the 1990s and 2000s, that many prostate cancers are so slow-growing they will never cause harm within a patient’s natural lifetime. The sensible response, it turned out, was often to watch carefully and do nothing. Active surveillance programmes formalised that insight, monitoring patients with regular PSA tests, MRI scans, and periodic biopsies to catch any tumours that might be changing character.
The problem is that biopsy, the only test that can definitively confirm whether a low-grade cancer has upgraded to something more threatening, is not a passive procedure. Inserting needles repeatedly into the prostate carries risks: pain, bleeding, infection. Some patients experience serious complications. And because the monitoring programmes run for years, or decades, each patient might face the procedure multiple times before their cancer does anything worth treating, or before they die of something else entirely. Getting better at predicting who actually needs a biopsy, and when, matters enormously.
Jeffrey Tosoian, an assistant professor of urology at Vanderbilt Health who led the study, framed the goal in direct terms. “For patients undergoing monitoring of low-grade prostate cancer, these findings suggest that use of the urine test can reduce the need for invasive biopsies without compromising prompt detection of higher-grade cancers that require treatment,” he said.
What the Numbers Actually Show
The test in question is called MyProstateScore 2.0 – Active Surveillance, or MPS2-AS. Like other so-called liquid biopsy approaches (a term the field uses, perhaps a bit loosely, to describe any test that draws clinical information from bodily fluids rather than tissue), it analyses molecular markers in urine that reflect what the prostate is actually doing at a cellular level. The active surveillance version was specifically calibrated for monitoring patients already diagnosed with Grade Group 1 cancer, the lowest and least aggressive tier in the prostate cancer grading system, and designed to detect the signs of upgrading to Grade Group 3 or above, the threshold at which most clinicians would recommend moving from watchful waiting to active intervention.
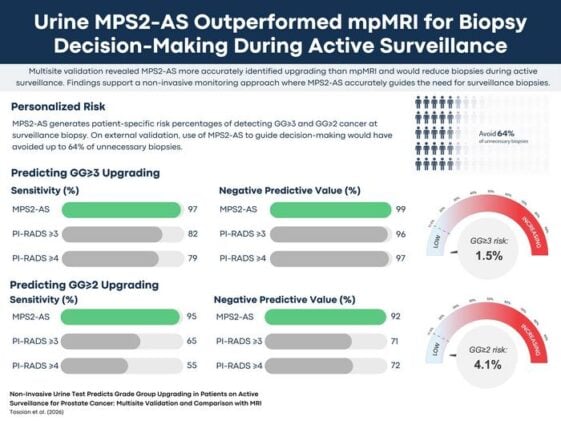
In the validation study, which enrolled 330 men already scheduled for a surveillance biopsy, MPS2-AS demonstrated a 99% negative predictive value for Grade Group 3 or higher disease, meaning that patients who tested negative had only around a 1 in 100 chance of harbouring high-grade cancer. The clinical implication is not subtle: for the vast majority of men who score negative, there is essentially no reason to proceed with the biopsy. Had the test been used to guide biopsy decisions in this cohort, it would have eliminated roughly 64% of what the researchers call “unnecessary biopsies” (those performed in patients who turned out not to have high-grade cancer) while missing only about 3% of the higher-grade tumours that do merit treatment.
MRI, which is currently the dominant noninvasive tool in active surveillance programmes, performed considerably worse. Using the standard PI-RADS scoring system and a threshold of 3 or higher to flag lesions for biopsy, MRI missed around 18% of Grade Group 3 upgrades and avoided fewer unnecessary procedures, sparing patients from roughly half of unnecessary biopsies rather than nearly two-thirds. The urine test also held up across subgroups, with consistent performance in Black patients, who are known to face higher prostate cancer incidence and mortality, as well as across both initial confirmatory biopsies and later surveillance biopsies.
There are caveats. The study drew its participants from 11 US practices, which is a reasonably broad validation sample but still not representative of every clinical setting or patient population. The field has seen promising biomarkers before, and some have stumbled when moved from controlled studies into messy routine care. Tosoian’s team also compared MPS2-AS against mpMRI using a specific PI-RADS threshold; a different threshold, or a different MRI interpretation protocol, might narrow the performance gap somewhat.
Still. What the data describes is a test that, in a carefully conducted multisite study, gave clinicians a practical tool they have not previously had: a reliable way to tell a significant proportion of men on active surveillance that they can skip their next biopsy without meaningful risk of missing something important. For a disease that affects millions of men globally and has historically been managed through years of low-grade procedural anxiety, that is not an incremental advance. Tosoian’s group has indicated that next steps include studying whether similar urine-based testing could detect cancer recurrence after treatment, extending the approach beyond surveillance into a broader slice of prostate cancer care.
The biopsy has been the irreducible minimum of prostate cancer monitoring for as long as the field has been trying to do it well. It may not be for much longer.
Source: Tosoian et al., The Journal of Urology (2026). doi:10.1097/JU.0000000000005095
Frequently Asked Questions
If I’m on active surveillance for prostate cancer, could I use this test instead of my next biopsy?
Possibly, though that decision would need to come from your urologist, not the test alone. MPS2-AS is still working its way into clinical guidelines, and the study that validated it was conducted across specialist urology practices rather than general settings. What the research does show is that men who test negative face only around a 1-in-100 chance of harbouring high-grade cancer, a level of certainty that many clinicians would find sufficient to defer a scheduled biopsy. Whether it’s right for your specific situation depends on your full clinical picture.
Why is prostate biopsy such a problem if it’s already standard practice?
Because active surveillance isn’t a one-time assessment; it runs for years, sometimes decades, meaning a patient might face the procedure repeatedly over their lifetime even if their cancer never progresses. Each biopsy carries a small but real risk of infection, bleeding, and pain, and the cumulative burden adds up. The search for a reliable noninvasive alternative has been underway for years, but until now no test had demonstrated the accuracy needed to confidently replace scheduled biopsies rather than merely supplement them.
Why does MRI currently miss some high-grade cancers that this urine test catches?
MRI shows anatomical structure: it can flag suspicious lesions based on their appearance and location in the prostate. But it can’t directly assess the molecular behaviour of cancer cells the way a biomarker test can. Some tumours that have upgraded to a more aggressive grade don’t look dramatically different on an MRI scan, at least not until they’ve grown considerably. A urine test that reads molecular signals from the prostate may pick up those cellular-level changes earlier, before they’re visible to imaging.
Is the 64% figure as good as it sounds?
It’s better than anything previously validated in this setting, but context matters. That figure refers to unnecessary biopsies avoided, meaning biopsies in patients who turned out not to have high-grade cancer on the actual biopsy. The test missed roughly 3% of the high-grade upgrades it was meant to catch, which is a low miss rate but not zero. Whether that 3% is acceptable clinically is a conversation between doctors and patients, not a number that speaks for itself.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.