Two major research advances are tackling the biggest challenges facing next-generation battery technology, potentially delivering safer and more durable power sources for everything from smartphones to electric vehicles. Scientists in Japan and Korea have developed innovative solutions that could make both sodium-ion and lithium-ion batteries more stable and long-lasting, addressing key obstacles that have limited their widespread adoption.
The breakthroughs focus on solving fundamental structural problems that cause batteries to degrade over time. By using copper doping and advanced mathematical frameworks, researchers have found ways to eliminate defects that previously shortened battery life and compromised safety.
Sodium-Ion Batteries Get Stability Boost
A team at Tokyo University of Science has solved a persistent problem plaguing sodium-ion batteries—microscopic defects called stacking faults that severely reduce performance. These structural flaws in β-NaMnO2 cathode materials cause rapid capacity loss during charging and discharging cycles.
Professor Shinichi Komaba and his team discovered that adding copper to the cathode material virtually eliminates these defects. “In a previous study, we found that among the metal dopants, Cu is the only dopant that can successfully stabilize β-NaMnO2,” explains Komaba. “In this study, we systematically explored how Cu doping can suppress SF and improve the electrochemical performance of β-NaMnO2 electrodes in Na-ion batteries.”
The results were dramatic. While undoped samples showed rapid capacity loss within 30 cycles, copper-doped versions demonstrated excellent stability, with some showing no capacity loss for over 150 cycles. At optimal copper levels of 12%, stacking fault concentration dropped to just 0.3%—a significant improvement from the 4.4% seen in lightly doped samples.
Lithium-Ion Safety Gets Mathematical Solution
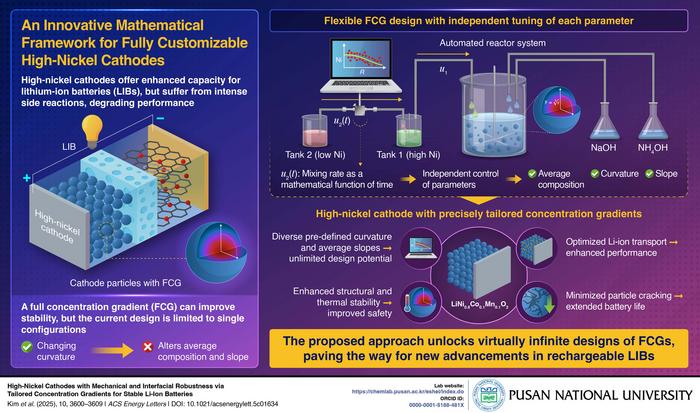
Meanwhile, researchers at Pusan National University developed a novel mathematical framework that enables precise control over lithium-ion battery cathode design. Their approach addresses safety concerns in high-nickel cathodes, which offer superior energy density but pose stability risks.
The team’s innovation allows independent control of multiple design parameters in concentration gradient cathodes. These structures feature nickel concentrations that gradually decrease from core to surface, replaced by more stable elements like cobalt and manganese.
“Unlike conventional methods, where adjusting one parameter affects the others, our approach allows independent and precise control over multiple descriptors, including average composition, slope, and curvature,” notes Associate Professor Hyun Deog Yoo, who led the research.
Key Performance Improvements
Both advances deliver significant performance gains:
- Sodium-ion batteries with copper doping showed no capacity loss over 150+ cycles
- Optimized lithium-ion cathodes retained 93.6% capacity after 300 cycles
- Enhanced mechanical stability reduces particle cracking
- Improved safety through better structural integrity
The sodium-ion breakthrough is particularly significant for cost-conscious applications. Sodium, the sixth most abundant element on Earth, offers much lower material costs than lithium while providing comparable performance when properly engineered.
“Our findings confirm that manganese-based oxides are a promising and sustainable solution for developing highly durable Na-ion batteries,” Komaba notes. “Owing to the relatively low cost of manganese and Na, this research will lead to more affordable energy-storage solutions for a variety of applications, including smartphones and electric vehicles.”
Real-World Impact
These developments address critical industry needs as battery demand skyrockets. The lithium-ion research involved collaboration between institutions in Korea and the United States, including the University of Illinois Chicago and Argonne National Laboratory, demonstrating the global priority placed on battery advancement.
The mathematical framework for lithium-ion cathodes uses automated reactor systems to synthesize materials with precisely controlled gradients. This level of customization was previously impossible with conventional two-tank coprecipitation methods, where adjusting one parameter automatically affected others.
Both research teams emphasize the broader implications for renewable energy adoption and grid storage applications. As electric vehicle sales accelerate and renewable energy installations expand, improved battery technology becomes essential for meeting sustainability goals.
The advances represent significant progress toward addressing supply chain vulnerabilities and performance limitations that have constrained battery technology deployment across multiple sectors.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.