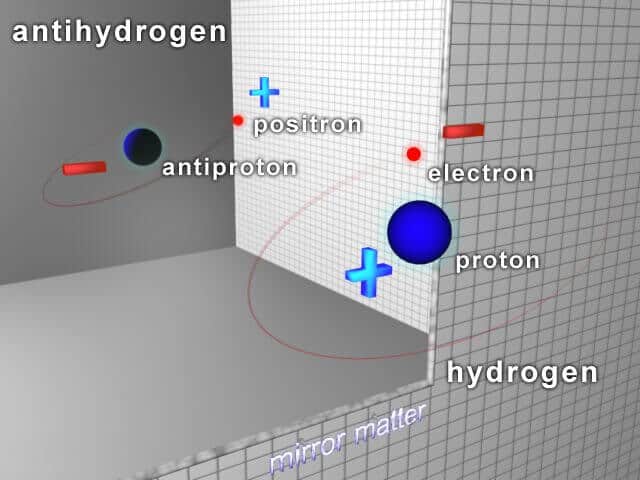
Antihydrogen is a particular kind of atom, made up of the antiparticle of an electron – a Positron – and the antiparticle of a Proton – an antiproton. Scientists hope that studying the formation of anti hydrogen will ultimately help explain why there is more matter than antimatter in the universe. In a new study published in EPJ D, Igor Bray and colleagues from Curtin University, Perth, Australia, demonstrate that the two different numerical calculation approaches they developed specifically to study collisions are in accordance. As such, their numerical approach could therefore be used to explain antihydrogen formation.
There are several methods of explaining anti-hydrogen creation. These involve calculating what happens when a particular kind of particle, made up of an electron and a positron bound together, called positronium, scatters on a proton or on an antiproton. The trouble is that devising numerical simulations of such collision is particularly difficult due to the presence of two centres for the occurrence: the atomic level with the proton and at the positronium level.
The authors employed two very different calculations – using a method dubbed coherent close-coupling – for both one- and two-centre collisions respectively in positron scattering on hydrogen and helium. Interestingly, they obtained independently convergent results for both approaches. Such convergence matters, as it is a way to ascertain the accuracy of their calculations for anti-hydrogen formation.
They then also compared the estimates of the area in the vicinity of the atom within which the positronium would need to be to ensure collision. They found excellent agreement with the two methods for hydrogen. However, their method did not prove quite as good for helium. This indicates that there is further room for improvement in the theory for helium before the approach can be applied to more complex atoms, such as magnesium and molecular hydrogen.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.