Every day, 22.5 billion gallons of sewage flow through America’s wastewater treatment plants.
And while these facilities clean water for environmental release, they also unwittingly release large amounts of carbon dioxide, a potent greenhouse gas, into nearby waterways and the air. Now, researchers at Johns Hopkins University have developed a new electrochemical process that captures carbon from treated water before it escapes, offering a surprising new way to fight climate change using existing infrastructure.
Electric Current Turns Wastewater Into a Climate Tool
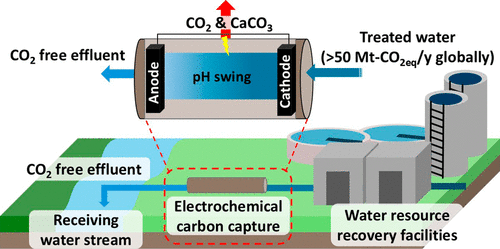
At the heart of the method is a compact device called an electrochemical cell. Co-developed by assistant professor Ruggero Rossi and graduate student Nakyeong Yun, the cell applies an electrical current to alter the pH of treated wastewater. This chemical tweak transforms bicarbonate ions—dissolved forms of carbon—into carbon dioxide gas and solid calcium carbonate. Both are then removed before the water is discharged into rivers and streams.
“We need to remove greenhouse gases from the atmosphere, and the easiest one to remove is CO2 because it’s most concentrated,” said Rossi, who teaches in the Department of Environmental Health and Engineering. “This is a way to leverage what we already have.”
First-of-Its-Kind Electrochemical Capture
In a study published in ACS ES&T Engineering, the researchers tested their carbon-capturing cell at the final stage of wastewater treatment—right before discharge. They used samples from four real-world treatment plants across the U.S., each with different water chemistry. Their goal was to measure how much inorganic carbon could be removed and how much energy the process would consume.
After 50 hours of continuous operation, the system proved stable and effective, though occasional cleaning was needed to manage mineral buildup. By adjusting variables like flow rate and electrode spacing, they improved performance without spiking energy costs.
Key findings include:
- More than 57% of dissolved inorganic carbon was captured
- 96% of that carbon was removed as gas, and 19% as solid carbonate
- Energy usage was as low as 3.4 kilowatt-hours per kilogram of CO2 captured
These results make the method competitive with current approaches used for air or ocean CO2 capture, but with one key advantage: wastewater infrastructure already exists in most cities.
A Retrofit With Major Impact
According to the researchers, implementing this system nationwide could prevent up to 12 million metric tons of CO2 emissions each year—about 28% of the total emissions from the U.S. wastewater treatment sector.
“This proof of concept study shows the potential of water reclamation facilities in contributing to a better environment,” said Rossi, “not only cleaning up contaminants from waste streams, but also removing greenhouse gases.”
Powered by Renewables, Built for the Real World
The approach isn’t without caveats. The carbon capture cells require energy to function, and to ensure the system remains carbon-negative, that power must come from renewable sources like solar or wind. Moreover, wastewater chemistry varies by location, time of day, and season, so some tailoring will be needed for widespread use.
Still, the researchers argue that the simplicity and adaptability of their design make it ideal for real-world deployment. Rather than building new facilities from scratch, cities could retrofit existing plants with this technology, integrating climate action directly into the water cycle.
Flush With Possibility
This project flips a long-overlooked environmental liability into an opportunity. By capturing carbon from treated water using a low-energy process and existing pipes, it could help municipalities shrink their climate footprints without breaking the bank—or digging new ditches.
“Before you see any of those plants chipping down the CO2 concentration in the atmosphere, it’s going to be a long time, and a lot of money,” Rossi said. “This is a way to leverage what we already have.”
Journal: ACS ES&T Engineering
DOI: 10.1021/acsestengg.5c00276
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.