In drug design and advanced materials, a molecule’s three-dimensional shape often determines everything. The same chemical formula can exist in mirror-image forms that behave completely differently. One might cure disease while its twin does nothing, or worse. For decades, chemists have struggled to control two types of molecular geometry at once: the handedness at specific atoms and the orientation of double bonds. Doing both typically required separate, complicated reactions that wasted half the starting material.
A team led by Professor Chuan He at Southern University of Science and Technology has now cracked this problem with a single catalytic system. Their rhodium-based catalyst simultaneously builds precise silicon chiral centers while dictating exact double-bond geometry, converting messy 50-50 mixtures into nearly pure products. The work, published in CCS Chemistry, demonstrates what chemists call enantioconvergent synthesis: funneling both mirror-image starting materials toward the same desired outcome rather than throwing away the “wrong” half.
A Chemical Traffic Controller
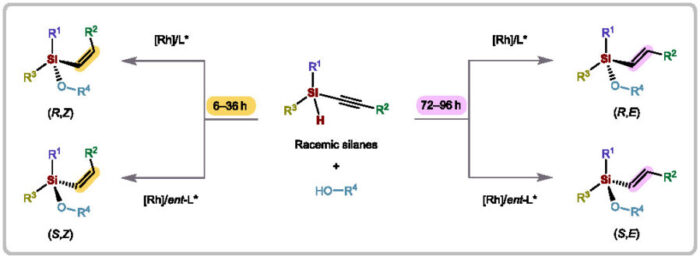
The breakthrough hinges on how the catalyst manages molecular handedness at silicon atoms while controlling whether carbon-carbon double bonds form in Z or E configurations. Traditional methods tackle these challenges separately, often requiring multiple steps and generating significant waste. He’s system does both jobs at once.
The reaction uses alkynyl monohydrosilanes—silicon compounds with a reactive hydrogen—and simple alcohols. Under rhodium catalysis at minus 20 degrees Celsius, both enantiomers of the starting material convert to a single product with up to 99 percent enantiomeric excess. By adjusting reaction time and temperature, the researchers can selectively produce any of the four possible stereoisomers: (R,Z), (R,E), (S,Z), or (S,E).
“Achieving switchable control over both Si and Z/E stereochemistry remains an elusive goal, especially for accessing all four stereoisomers from the same starting materials,” He explains.
Chloride ions turned out to be essential. Mechanistic studies revealed these ions enable inversion at silicon centers, flipping “wrong” configurations into the desired arrangement. An alkenyl-rhodium intermediate activates the silicon-hydrogen bond, while the chiral ligand on the catalyst guides everything toward the correct three-dimensional structure. Short reactions at low temperatures favor the kinetically preferred Z products, which are typically harder to make because they’re less stable. Longer reaction times allow controlled conversion to the more common E forms.
Practical Versatility
The team tested the method on diverse substrates, from simple chains to bulky, complex structures. The reaction maintained high selectivity across this range, working with various alcohols and functional groups without losing precision. Yields stayed good to excellent throughout, and the process operates with 100 percent atom economy—every bit of starting material ends up in the product.
The resulting vinyl silyl ethers can be transformed into other silicon-containing compounds without losing their carefully constructed geometry. This matters because silicon-based molecules appear throughout modern chemistry, from pharmaceutical candidates to materials for electronics and catalysis.
Density functional theory calculations confirmed the experimental observations, showing how the catalyst orchestrates multiple stereochemical decisions simultaneously. Rather than sequential compromises, the system treats dual stereochemical control as a coordinated design problem.
The implications extend beyond silicon chemistry. By proving that a single catalyst can manage multiple types of molecular geometry at once, the work suggests new strategies for synthesizing other complex molecules more efficiently. Future efforts will likely explore whether similar unified approaches can control stereochemistry in carbon-carbon bond formation or other challenging transformations where molecular shape determines function.
CCS Chemistry: 10.31635/ccschem.025.202506802
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.