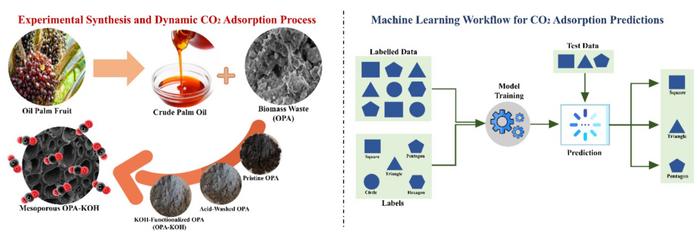
In a Penang laboratory, researchers have figured out how to turn one environmental problem into a solution for another. Oil palm ash, the residue left behind when palm kernel shells burn in Malaysian mills, can capture carbon dioxide from the air with surprising efficiency.
The material performs this trick despite having a surface area far smaller than most commercial carbon-capture products. Dr. Azam Taufik Mohd Din and his team at Universiti Sains Malaysia treated the ash with acid, heated it to 700 degrees Celsius, and activated it with potassium hydroxide. The result: a mesoporous structure that grabbed 2.9 millimoles of CO2 per gram of material.
That capacity rivals products engineered with much larger surface areas. The secret lies not in size but in architecture. With an average pore diameter of 72.71 angstroms, the material creates channels perfectly sized for CO2 molecules to enter and stick.
This is not just recycling, it is upcycling at the molecular level. We are taking a waste product that often ends up in landfills and turning it into a high-performance tool for carbon capture.
Machine Learning Meets Molecular Design
The research, published August 18 in Carbon Research, breaks new ground by pairing laboratory experiments with artificial intelligence. Traditional models of adsorption behavior often miss the complexity of how gases interact with porous materials. Mohd Din’s team deployed machine learning algorithms to predict CO2 capture under different conditions.
A bilayered neural network achieved an R-squared value exceeding 0.99, meaning it predicted experimental outcomes with near-perfect accuracy. The model could simulate changes in temperature, flow rate, and gas concentration faster than running physical tests.
This shows ML is not just a trend, it is becoming essential. It allows us to simulate, optimize, and understand adsorption processes faster and more deeply than ever before.
The thermodynamic analysis revealed that CO2 sticks to the material primarily through physisorption, weak physical forces that work best at lower temperatures. At 30 degrees Celsius, the material performed optimally. Raising the temperature to 60 degrees cut the capture capacity nearly in half, as molecules gained enough energy to escape the surface.
From Agricultural Waste to Climate Technology
Malaysia produces more than 20 million tons of palm oil annually, generating vast amounts of ash that presents disposal challenges. Converting this waste into a carbon-capture material offers a closed-loop solution. The production process requires less energy than synthesizing commercial adsorbents from scratch.
The material’s performance depends heavily on the ratio of ash to potassium hydroxide. At a 1:2 ratio, pore development reached an optimum. Push the ratio to 1:3, and the carbon structure begins to collapse from excessive activation. The research team tested different ratios systematically, finding that too little activation left performance gains on the table while too much destroyed the useful architecture.
Flow rate mattered too. At 30 milliliters per minute, CO2 molecules had enough contact time with the adsorbent surface. Double the flow rate to 60 milliliters per minute, and breakthrough occurred prematurely. The molecules simply moved too fast to stick effectively.
Higher CO2 concentrations improved capture, up to a point. At 15 percent CO2, the material reached its maximum capacity. But at 12 percent, something unexpected happened: the breakthrough curve flattened, suggesting an optimal balance between adsorption rate and gas flow that extended capture time.
The research positions Universiti Sains Malaysia’s School of Chemical Engineering as a regional hub for waste valorization and AI-driven materials science. The team plans pilot-scale tests in industrial settings: flue gas treatment, biogas upgrading, direct air capture systems. Long-term stability tests will determine whether the material can survive repeated regeneration cycles without losing effectiveness.
Compared to other biomass-derived adsorbents in the literature, oil palm ash holds its own. Some studies report higher surface areas, but surface area alone does not determine capture efficiency. Pore architecture, selectivity, and functionalization matter just as much. The mesoporous structure of the Malaysian material enables efficient CO2 diffusion and faster adsorption kinetics.
The integration of machine learning into materials development could accelerate the search for better adsorbents. Rather than testing hundreds of conditions in the laboratory, researchers can use predictive models to narrow the parameter space, then validate the most promising candidates experimentally. This approach reduces both time and cost.
With climate targets tightening worldwide, affordable and scalable carbon capture technologies face urgent demand. This research delivers both: a low-cost, high-efficiency adsorbent born from agricultural waste, guided by computational prediction. The material works, the economics look favorable, and the raw material supply presents no shortage.
So the next time palm oil appears in your food or cosmetics, remember: from the same industry’s waste stream, scientists are engineering materials to pull carbon dioxide from the atmosphere, one molecule at a time.
Carbon Research: 10.1007/s44246-025-00227-3
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.