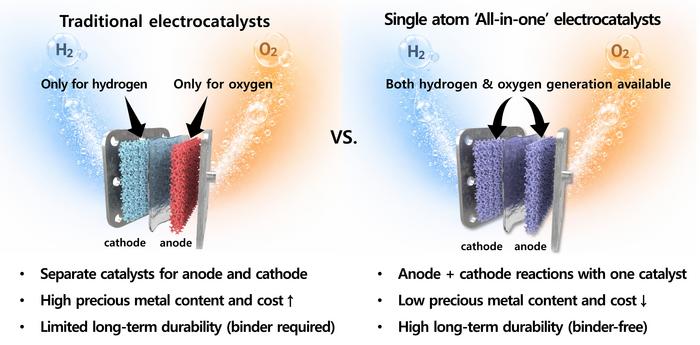
At the Korea Institute of Science and Technology in Seoul, a cluster of researchers has been wrestling with one of the more awkward constraints in green hydrogen chemistry. To split water into its constituent gases, you need two separate reactions running simultaneously — one at the cathode releasing hydrogen, another at the anode releasing oxygen — and they have, until recently, demanded two entirely different types of catalyst to drive them. That redundancy costs money, complicates electrode design, and burns through iridium and platinum at rates that make commercial-scale deployment look like a distant ambition.
The solution the KIST team arrived at sounds almost implausibly tidy: a single catalyst that handles both reactions at once, built from individual iridium atoms dispersed so finely across a metal support that you need an electron microscope to confirm they’re actually there.
Water electrolysis is, on paper, a simple proposition. Apply electricity to water and it splits. Hydrogen bubbles off one electrode, oxygen from the other. Powered by solar or wind, the process produces green hydrogen without a carbon atom in sight. The catch is the chemistry at each electrode. The hydrogen evolution reaction, or HER, and the oxygen evolution reaction, OER, are governed by fundamentally different kinetics. Alkaline conditions — the cheapest operating environment — slow the hydrogen side down by two to three orders of magnitude compared with acidic systems, because the water molecule must first be cracked apart before hydrogen can be released. The oxygen side demands a four-step electron transfer through multiple intermediate compounds, requiring overpotentials exceeding 300 millivolts even in state-of-the-art systems. Platinum handles the hydrogen reaction well. Iridium oxide manages the oxygen side. Neither does both adequately, and you need substantial quantities of each.
Which is precisely what makes the KIST result, published in Advanced Energy Materials, worth paying attention to.
The catalyst goes by the designation Ir@Mn-Ni-PA. The scaffolding is a layered double hydroxide — a class of materials with sheet-like structures and tunable chemistry — built from nickel and manganese. Phytic acid, a naturally occurring compound from plant seeds, is used to modify the surface. Its phosphate groups act as anchoring points, chemically grabbing iridium ions from solution and holding them in place individually, one atom at a time, during a mild hydrothermal synthesis. The result is a hierarchical nanoarray — a porous, wettable structure with iridium atoms separated from their neighbours by distances measured in ångströms.
What makes individual atoms useful, rather than clusters or nanoparticles, is that every single atom becomes an active site rather than being buried inside inert bulk material. A lump of iridium uses most of its atoms wastefully. Spread the same amount across a surface atom by atom, and each one can participate in the reaction. The KIST team’s catalyst contains iridium at just 1.42 atomic percent — a fraction of the loading in conventional precious-metal electrodes.
The numbers from the electrochemical testing are striking. For HER, Ir@Mn-Ni-PA required only 65 millivolts of overpotential to reach a benchmark current density of 10 milliamps per square centimetre — close to the 47 millivolts needed by commercial platinum-carbon catalysts, and far below the 302 millivolts demanded by the plain nickel-manganese support without iridium. For OER, it achieved 272 millivolts, outperforming commercial ruthenium dioxide, which needed 364 millivolts. More telling still is what happened at higher currents: at 100 milliamps per square centimetre for HER, Ir@Mn-Ni-PA needed only 132 millivolts, while platinum-carbon required 201 millivolts and gave up. Industrial electrolysers must operate at high current densities to be economically viable, and that is precisely where conventional catalysts tend to fall apart.
Understanding why the catalyst works required computational chemistry. Density functional theory calculations revealed that incorporating iridium atoms narrows the bandgap of the nickel-manganese support, improving electron conductivity across the material. More subtly, charge redistributes around the iridium sites — the d-band centre of the iridium atoms shifts further from the Fermi level than in metallic iridium clusters. That shift matters because of a principle known as the Sabatier principle: a catalyst binds its reaction intermediates neither too strongly nor too weakly. Metallic iridium binds hydrogen too firmly. The modified electronic environment in Ir@Mn-Ni-PA produces a near-optimal hydrogen adsorption free energy of -0.06 electron-volts, almost exactly zero, which is the theoretical sweet spot for HER.
The manganese dopant plays its own role. It adjusts the adsorption energies of the oxygen-containing intermediates in OER, while its reversible redox behaviour helps generate a catalytically active nickel oxyhydroxide phase under operating conditions. In situ Raman spectroscopy confirmed this surface transformation at work — above 1.3 volts, the nickel hydroxide in the catalyst converts to nickel oxyhydroxide, the species actually doing the oxygen chemistry. The iridium atoms enhance this process through electronic coupling with the support, rather than acting as independent catalytic centres.
The phytic acid modification also makes the electrode surface dramatically more water-loving. Contact angle measurements showed the surface going from 79 degrees — modestly hydrophilic — down to 12 degrees after phytic acid treatment. That near-superhydrophilic surface pulls electrolyte deep into the porous structure and helps gas bubbles detach cleanly, preventing the blockage that degrades performance at high current densities.
Durability is where hydrogen catalysts typically fail. The KIST team tested the catalyst in an anion exchange membrane water electrolyser — a device architecture that combines the efficiency of proton exchange membrane systems with the cost advantages of alkaline chemistry — under industrially relevant conditions for 300 hours. The electrode maintained stable performance throughout, with a voltage decay rate of just 77 microvolts per hour. Notably, the cell actually improved over the test period: after 300 hours, it was delivering 2.03 amps per square centimetre at 2.0 volts, up from 1.64 amps at the start, as the catalyst continued to activate. Commercial platinum-carbon electrodes lost nearly 40 percent of their current within 28 hours under comparable conditions.
“This work is highly significant as it resolves the two essential reactions for hydrogen production using a single catalyst while reducing precious metal consumption,” said Dr Na Jongbeom, who led the research. “This technology will accelerate the commercialisation of water electrolysis devices and provide substantial support for expanding hydrogen energy.”
A key engineering decision contributed to the performance gains. Rather than mixing the catalyst with a polymer binder and coating it onto an electrode, the team grew the material directly onto nickel foam supports during synthesis. Binders block active sites and delaminate under prolonged operation; the direct-growth approach eliminates both problems while improving electrical contact between the catalyst and the substrate. When the team compared direct-growth against spray-coated powders in actual electrolyser cells, the performance advantage was substantial.
Green hydrogen remains expensive compared with fossil-derived alternatives, and catalyst cost is a meaningful fraction of that gap. The precious-metal loading in current commercial electrolysers represents both a financial constraint and a supply chain vulnerability — iridium is one of the rarest elements in the Earth’s crust. A catalyst architecture that delivers competitive or superior performance at a fraction of the metal loading addresses both problems simultaneously. Whether Ir@Mn-Ni-PA can survive the transition from laboratory cells to full-scale industrial stacks — with their higher pressures, variable loads, and years-long operational demands — remains to be demonstrated. But the underlying principle, that atomic-level engineering can extract more function from less material, points toward a hydrogen economy that might actually be affordable to build.
Study link: https://advanced.onlinelibrary.wiley.com/doi/10.1002/aenm.202506645
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.