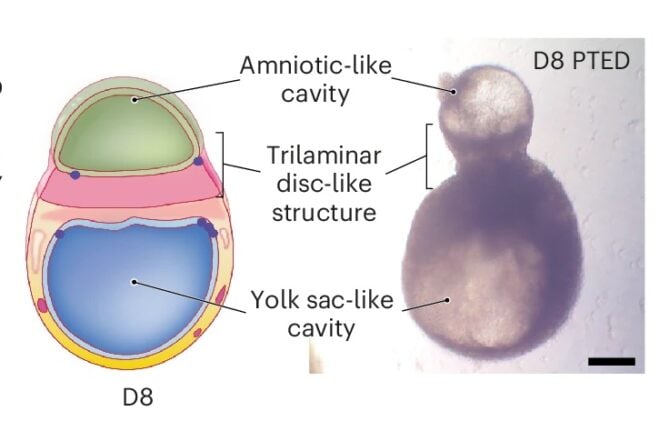
Shiyu Sun was looking at a small blob of human stem cells on the eighth day of an experiment. At one end of the blob, a cavity. At the other end, another cavity. Between them, a three-layered disc. The thing shouldn’t have been there: according to the canonical account of early human development, the structure at the ventral end, which looked for all the world like a yolk sac, is supposed to come from a completely different cell type than the one Sun had started with. He checked again. It still looked like a yolk sac.
That observation, now published in Nature Cell Biology by a team at the University of Michigan working with colleagues at the Chinese Academy of Sciences, is the first time a stem cell model of a human embryo has produced a yolk-sac-like structure spontaneously, without genetic manipulation, from a single starting population of primed pluripotent stem cells. It is both a methodological milestone and a small puzzle for the developmental biology textbooks.
Why the Yolk Sac Matters
A yolk sac is the early embryo’s improvised pantry. It forms while the placenta is still being assembled, stores nutrients, and hosts the first blood cells the body will ever produce. In humans it is understood to arise from the hypoblast, a distinct cell layer that sits beneath the epiblast in the days after implantation. Labs trying to model this phase of development have either forced the issue with gene editing, inducing cells to behave like hypoblast derivatives, or have added hypoblast-like cells as ingredients in a mix. The Michigan team did neither.
What they did do was patterning. Sun and his colleagues, led by Jianping Fu, stamped circular adhesive islands onto glass coverslips using a rubber-like polymer stamp. Each island was 800 micrometres across, which happens to be roughly the diameter of the human epiblast disc at the moment gastrulation begins. Primed stem cells seeded onto these patches were nudged with BMP4, a signaling molecule that in a normal embryo comes from surrounding extraembryonic tissues but is absent in this setup. The cells began gastrulating. And then, unprompted, they built more than anyone had expected.
Surprise at Day Eight
What started as a flat disc thickened into three cellular layers: an ectodermal precursor on one side, an endodermal precursor on the other, and a mesodermal layer in between. A cavity lined with amnion-like cells opened on the dorsal side. On the opposite side, a second cavity formed, this one surrounded by a thin layer of endoderm-like cells that expressed HNF4A, a gene that in real embryos marks yolk sac identity. Tissues within the ventral compartment began producing blood cells. In timing and composition, the Day 8 structure mapped onto human development at roughly 16 to 21 days after fertilization, a window almost nobody can study directly.
“It was pretty surprising to find these yolk-sac-like structures,” Sun said. His first instinct had been to dismiss the interpretation; it simply wasn’t on the cards for the kind of starting cells the experiment used.
To confirm the identity of what they were looking at, the team turned to lineage tracing. They engineered the cells so that any cell which activated the gastrulation marker TBXT would permanently switch on a green fluorescent protein. Cells that had passed through that gastrulation checkpoint were, from then on, tagged. When the team examined the yolk-sac-like compartment at Day 8, nearly every mesodermal cell surrounding it was green, and roughly half of the endoderm cells lining the sac were green too. The cells had, in other words, gone through gastrulation and then become extraembryonic.
That wasn’t supposed to happen.
“According to canonical knowledge of human development, the yolk sac should come from hypoblast cells,” Fu said. The inference the authors draw, carefully, is that primed epiblast cells in the peri-gastrulation embryo may retain an extraembryonic potential that nobody had credited them with, and may be able to route themselves into yolk sac lineages through a back door that textbooks don’t acknowledge. If true in vivo, it complicates a piece of developmental biology that had felt settled for decades.
What It Can and Can’t Tell Us
The model has real limits. The three germ layers it forms are thicker than they should be. It lacks trophoblast cells, so it cannot develop a placenta and cannot be taken to be a complete model of the embryo. Somewhere between 15% and 20% of the cultures form the full structure, depending on which stem cell line goes in, and efficiency drops off sharply after Day 8 as the embryoids detach from their plates and drift loose.
Even imperfect models of peri-gastrulation are useful, though. A large fraction of pregnancies end before the person carrying them knows they are pregnant, and a good share of that loss happens in exactly this window. What goes wrong is poorly understood, partly because post-implantation human embryos are unavailable for study for ethical and practical reasons, and partly because the cell lineages involved are genuinely difficult to reach. A dish that reliably grows a trilaminar disc, an amnion, a yolk sac and the first haematopoietic islands gives researchers something to probe.
Whether epiblast cells in the actual human embryo do what they did in the Michigan dish is an open question, and one that, given the 14-day rule limiting how long human embryos can be cultured for research, may stay partly open for a long time. But the Fu team’s result quietly suggests that early human cells may be stranger, and more flexible, than the canonical diagram allows. The road between the flat disc and the first blood cell is not the one-way street it looks like in a cross-section.
DOI: 10.1038/s41556-026-01930-y
Frequently Asked Questions
How is this stem cell model different from other embryo models?
Earlier models of post-implantation human development have generally needed either genetic manipulation (inserting transcription factors to force cells into extraembryonic identities) or mixing in multiple cell types from different pluripotency states to build structures like the yolk sac. This one starts from a single population of ordinary primed pluripotent stem cells, uses only a physical pattern and a standard signaling molecule, and still ends up with a trilaminar disc, amnion-like cavity, and yolk-sac-like structure. That makes it cleaner, easier to replicate, and less ethically fraught.
Could this model help explain early miscarriage?
Potentially, yes. A large share of pregnancy losses happen in the first few weeks after fertilization, before many women know they are pregnant, and this model spans roughly the 16-to-21-day window when a lot of that failure occurs. Having a reproducible dish-based system that recreates germ-layer formation, amnion emergence and early blood-cell generation gives researchers a way to screen what goes wrong at those stages without working with real human embryos. It isn’t a complete model, but it is a useful one.
What does the 14-day rule have to do with this research?
The 14-day rule is a longstanding international guideline, now loosened in some countries, that limits the culture of actual human embryos in research to fourteen days past fertilization. Because much of gastrulation and yolk sac formation happens after that point, researchers cannot study these events directly in human embryos. Stem cell-based models like this one are designed to sit in that gap, offering an ethically and practically accessible way to probe a developmental window that is otherwise closed to direct observation.
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.