Key Takeaways
- Recent research shows that intestinal epithelial cells in multiple sclerosis patients activate immune functions, leading to brain-related inflammation.
- This study changes the perspective on MS origins, suggesting that the gut microbiome’s role is more significant than previously thought.
- Elevated MHC class II expression in gut epithelial cells may present antigens that trigger aggressive immune responses, linked to MS.
- Targeting the gut microbiome could provide new treatment avenues for MS, shifting from traditional immune suppression methods.
- Further research is needed to clarify the link between gut bacteria and MS, as current human data is limited but promising.
Something is happening in the small intestine long before the first nerve fibre starts to fray. Lining the gut wall, a layer of epithelial cells quietly absorbs nutrients, manages the microbial population pressing against them from the lumen, and in ordinary circumstances, minds their own business. They are not supposed to be involved in the immune system’s more consequential decisions. That is the job of dedicated immune cells, the professionals. But in people with multiple sclerosis, it seems the gut’s epithelial layer has taken matters into its own hands, switching on molecular machinery it has no business operating and using it to prime a particularly destructive category of immune cell. Those cells then travel to the brain.
The finding, published this week in Science Immunology by researchers at Keio University in Japan, reshapes how scientists think about MS origins. Multiple sclerosis has long been understood as a disease of the central nervous system: immune cells crossing the blood-brain barrier and stripping away the myelin sheaths that insulate nerve fibres, causing the neurological disruption that leaves roughly 2.9 million people worldwide with escalating disability. The immune attack is real, but the question of where it starts may have had the wrong answer all along.
The gut microbiome has been circling this question for a few years now. Patients with MS show characteristic alterations in their gut bacteria populations, and researchers working with experimental autoimmune encephalomyelitis (EAE), the standard mouse model of MS, found that eliminating gut microbes with antibiotics substantially reduced disease severity. Microbial metabolites also seemed to nudge immune cells toward inflammatory profiles. Gut microbes were clearly relevant. But how gut signals were being converted into brain damage, at the cellular level, was genuinely murky. As Tomohisa Sujino, who led the research group, noted, “the mechanisms linking gut microbes, intestinal immunity, and brain inflammation remain unclear.”
Not exactly, but the new research suggests the gut plays a much more active role in triggering MS than previously recognised. Intestinal epithelial cells appear to switch on antigen-presenting machinery during the disease, priming a class of aggressive immune cell that then travels to the brain and spinal cord. Whether this gut process is the original trigger or an amplifying step in a longer chain is still being worked out.
The leading hypothesis involves molecular mimicry: gut bacteria may carry surface proteins that structurally resemble myelin, the insulating material around nerve fibres that MS destroys. When intestinal epithelial cells present bacterial fragments to immune cells using MHC II machinery, the resulting response may inadvertently target the brain. The antibiotic experiments in this study support the idea that microbiota-derived antigens are involved, though which specific bacteria are responsible is unknown.
The study raises that possibility more seriously than before. Current MS therapies mainly focus on suppressing immune activity or targeting B cells, with the gut receiving little attention. If epithelial antigen presentation in the small intestine is a key initiating step, then interventions that alter the gut microbiome or reduce the epithelium’s antigen-presenting activity could offer a new therapeutic angle. Diet, probiotics, or more targeted microbiome interventions are among the candidates worth exploring.
The human data in this paper are promising but limited: intestinal biopsies from four MS patients showed a consistent pattern of elevated epithelial MHC II expression and Th17 cell accumulation compared with three healthy controls. The mouse work is more extensive and mechanistically detailed. Larger human cohort studies are needed to confirm how consistent this gut signature is across the MS population and whether it correlates with disease severity or progression.
His team’s answer turned on a molecule called MHC class II. Normally, MHC II is displayed on the surface of dedicated antigen-presenting cells, the body’s professional educators, which use it to show fragments of foreign proteins to naive T cells and tell them what to attack. Intestinal epithelial cells, the ordinary lining of the gut, do not typically do this. Or so everyone assumed.
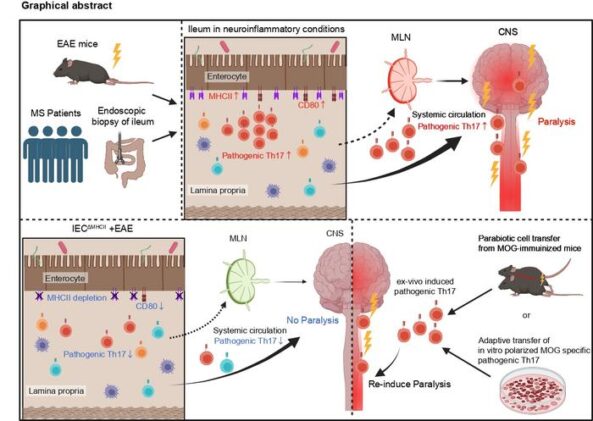
When Sujino’s group performed single-cell RNA sequencing on intestinal biopsy samples from MS patients and healthy controls, they found that enterocytes, the workhorse absorptive cells of the small intestine, were running an elevated antigen presentation programme in patients with MS. The gene CIITA, which acts as a master switch for MHC II expression, was firing in epithelial cell clusters where it had no expected business. Antigen presentation scores in the intestinal lining of MS patients were significantly higher than in healthy individuals, while scores in professional immune cells showed no particular difference between groups. The epithelium was doing something unusual. The professional antigen presenters were not.
In EAE mice the pattern repeated. MHC II expression on epithelial cells of the small intestine rose sharply as disease developed, most dramatically in the ileum, and this increase appeared alongside an accumulation of Th17 cells. Th17 cells are a particular subspecialty of the immune system’s attack force. They produce cytokines that are good at driving inflammation and, crucially, have a well-documented role in MS. The gut was full of them.
To test whether the epithelial cells were doing anything functional with their newly acquired machinery, the team generated mice in which MHC II could be selectively deleted from intestinal epithelial cells while remaining intact in conventional immune cells. These IECΔMHCII mice, as they are termed in the paper, developed substantially milder EAE when given the standard immunisation protocol. The pathogenic Th17 population in their spinal cords was significantly reduced. Depletion of dendritic cells, the professional antigen presenters, did not produce the same effect. Whatever was generating the problematic Th17 cells in the gut, it was the epithelial layer, not the trained specialists.
The migration question was perhaps the most technically elegant part of the study. Using a transgenic mouse line carrying the Kaede protein, which fluoresces green by default but shifts permanently to red when hit with violet light, the researchers could illuminate a small patch of the ileum and then watch where the marked cells went over the following 48 hours. They found that photoconverted CD4 T cells from the intestinal lamina propria accumulated in the spinal cord during the disease phase. A subset of these cells, carrying the surface marker CXCR6 which flags them as particularly encephalitogenic, preferentially exited the gut layer and showed up in the central nervous system. Gut-primed cells were making the journey. The team also ran parabiosis experiments, surgically connecting pairs of mice with shared blood supply, to show that MHC II expression in the immunised animal was what mattered for disease induction; the partner’s MHC II status was largely irrelevant. Priming was happening locally, in the gut epithelium, not downstream.
Antibiotic treatment complicated the picture in an interesting way. Broad-spectrum antibiotics reduced EAE severity and cut the proportion of myelin-reactive Th17 cells in the gut, but left MHC II expression on intestinal epithelial cells essentially unchanged. This suggests the epithelial cells are probably presenting microbial antigens, some of which may cross-react with myelin through molecular mimicry, rather than presenting myelin fragments directly. What the gut bacteria are putting in the display case, and exactly which bacterial species are responsible, remains an open question.
Clinically, MS treatment has mostly targeted B cells, or broadly suppressed immune activity, with the gut hardly featuring in the therapeutic picture. Shohei Suzuki, who co-led the study with Sujino, was direct about what the findings imply: “our study highlights the gut as an important therapeutic site.” Interventions aimed at altering the gut microbiome, or at dampening the antigen-presenting activity of intestinal epithelial cells, could represent a new front in MS treatment. Diet and microbial composition are, in principle, more tractable targets than the immune cascade they eventually unleash.
Whether the specific bacteria or the precise antigens involved in human MS can be pinned down is the next major challenge. The patient cohort in this study was small, four MS cases against three healthy controls, enough to establish a pattern but not to characterise its boundaries. What is clear is that the gut is not a passive bystander in autoimmune brain disease. Its lining cells are running a parallel immune training programme, and in MS, that programme appears to be producing graduates with the wrong target.
DOI / Source: https://doi.org/10.1126/sciimmunol.aec1627
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.