Iron pills often fix the blood yet punish the gut. A new lab-built combo aims to do both jobs cleanly, and it just passed early tests in mice reported in a peer reviewed journal from the American Chemical Society.
Researchers in India engineered a synbiotic iron delivery system that packages an iron complex with a probiotic and its favorite food inside a mucoadhesive hydrogel. In anemic mice, two weeks of dosing restored hemoglobin, quieted inflammatory signals in the colon, and brought gut microbes back toward a healthier balance. The work, published in ACS Applied Materials & Interfaces, suggests a path to treat iron deficiency anemia while protecting the intestine rather than irritating it.
If you have ever opened a standard iron tablet, you know the look and smell: a chalky disk that can sit heavy in the stomach. This new material acts more like a soft, protective raft. Picture a clear gel bead, no bigger than a seed pearl, that clings to the intestinal lining and releases its cargo in a controlled way as it slides along the wet folds of the gut.
What The New Formulation Does Differently
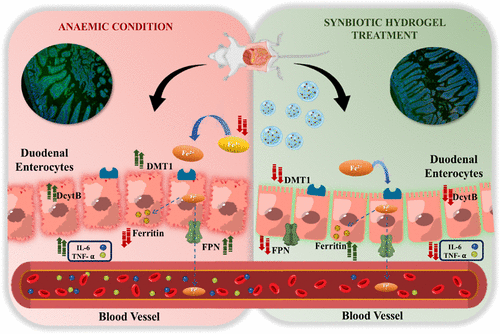
The team combined three parts into one: an iron dextran complex for replenishing blood iron, live Lactobacillus rhamnosus as a beneficial microbe, and soluble dietary fiber from millet to feed that microbe. All three were encapsulated in a thiolated hyaluronic acid hydrogel designed to adhere to mucus and meter release. In cell tests, the construct was biocompatible. In animals, it did more than raise hemoglobin. It also shifted molecular markers in a direction consistent with restored iron handling and reduced inflammation.
The investigators tracked transcripts tied to iron transport, including DMT1 and DcytB, and observed normalization after treatment. Ferritin protein rose, indicating replenished stores. Colon tissue showed lower TNF alpha and IL 6 by both immunofluorescence and Western blot. Fecal iron levels resembled non anemic controls, a sign that more iron was taken up rather than left to irritate the lumen. Microbiome readouts moved toward baseline as well. These are mouse data, not human outcomes, yet the pattern addresses the core tradeoff clinicians see with conventional iron salts: effective systemic repletion at the cost of local gut disruption.
“By advancing biomaterial-based iron delivery, this research offers a transformative approach to address anemia, directly contributing to improved nutrition and long-term public health.”
That claim will need human testing to stand. Still, the engineering logic is sound. Encapsulation can shield sensitive microbes and time the release of both iron and prebiotic substrates. Mucoadhesion can keep the payload where transporters live. Pairing a probiotic with its preferred fiber can tilt the local ecology away from pro inflammatory states that free iron can promote.
Why It Matters For Patients And Public Health
Iron deficiency anemia remains common worldwide across ages and settings. Oral iron is cheap and scalable, but side effects drive poor adherence. A formulation that improves absorption while reducing gut irritation could raise real world effectiveness. It could also reduce the need for higher doses, which tend to worsen gastrointestinal complaints. From a systems view, the inputs are familiar, the lever is delivery, and the output is a better balance between efficacy and tolerability.
There are limits. Mouse intestines are not human intestines. Regulatory paths for live biotherapeutic products can be complex. Stability during manufacturing and storage must be proven. Dosing schedules and interactions with antibiotics or existing probiotics need study. Cost will matter, too. Even so, the approach integrates known parts in a coherent design, which often translates faster than invention from scratch.
“The findings suggested a step toward the development of an integrated oral iron supplementation medium with improved bioavailability and reduced gut inflammation.”
Next steps should include randomized, controlled human trials that measure not only hemoglobin recovery and ferritin but also stool iron, calprotectin, patient reported gastrointestinal symptoms, and microbiome composition over time. Head to head comparisons with standard ferrous sulfate would clarify value. If results hold, this could be an incremental but meaningful upgrade to a therapy used by millions.
For now, the study offers a clear takeaway: delivery details matter. By tuning the microenvironment at the mucosal surface, and by pairing iron with allies rather than leaving it to react freely, it may be possible to nourish blood and spare the gut at the same time.
ACS Applied Materials & Interfaces: 10.1021/acsami.5c11368
ScienceBlog.com has no paywalls, no sponsored content, and no agenda beyond getting the science right. Every story here is written to inform, not to impress an advertiser or push a point of view.
Good science journalism takes time — reading the papers, checking the claims, finding researchers who can put findings in context. We do that work because we think it matters.
If you find this site useful, consider supporting it with a donation. Even a few dollars a month helps keep the coverage independent and free for everyone.